Navigating SCADA System Challenges: Enhancing Reliability and Efficiency with Comprehensive Support Services
 SCADA systems are crucial for manufacturing and industrial operations, serving as the vital link between control systems and human oversight. But they’re complex and vulnerable to issues like technical reliance, communication disruptions, and cyber threats. When SCADA fails, production stops—prompt recovery is vital. Discover how structured SCADA support and improvement, including partnering with external experts, can prevent disruptions.
SCADA systems are crucial for manufacturing and industrial operations, serving as the vital link between control systems and human oversight. But they’re complex and vulnerable to issues like technical reliance, communication disruptions, and cyber threats. When SCADA fails, production stops—prompt recovery is vital. Discover how structured SCADA support and improvement, including partnering with external experts, can prevent disruptions.Read More
Improving Performance of MES through Support
 Discover how MES support services are enhancing operational performance in manufacturing. Explore Avanceon’s insights into optimizing MES systems for increased efficiency and productivity.
Discover how MES support services are enhancing operational performance in manufacturing. Explore Avanceon’s insights into optimizing MES systems for increased efficiency and productivity.Read More
Optimizing Performance During Production
 Learn how we helped a customer bring their batch time down from 60 hours to under 24 hours all while maintaining 100% production during the changes. By using Machine Learning and AI in the project, the result of the required upgrade cut the cycle time by more than half, on a batch worth $500,000. Quite an impact to the bottom line.
Learn how we helped a customer bring their batch time down from 60 hours to under 24 hours all while maintaining 100% production during the changes. By using Machine Learning and AI in the project, the result of the required upgrade cut the cycle time by more than half, on a batch worth $500,000. Quite an impact to the bottom line.Read More
Delivering Results in a Challenging Project: Two Customers, One Project
 Learn how Avanceon handled navigating two customers during a single large greenfield project. While the EPC was technically our customer, we had an existing 20+ relationship with the manufacturer. Situations such as these have the potential to end badly, however, we were able to accommodate everyone and left with two pleased customers.
Learn how Avanceon handled navigating two customers during a single large greenfield project. While the EPC was technically our customer, we had an existing 20+ relationship with the manufacturer. Situations such as these have the potential to end badly, however, we were able to accommodate everyone and left with two pleased customers.Read More
The Ins and Outs of OT Innovation in a New Plant
 Learn the key control, SCADA, and batch system steps and considerations when implementing a greenfield manufacturing facility from the ground up. Manage risk and drive innovation from preliminary design to production support and training.
Learn the key control, SCADA, and batch system steps and considerations when implementing a greenfield manufacturing facility from the ground up. Manage risk and drive innovation from preliminary design to production support and training.Read More
Upgrades and Commissioning in a Validated world – Looking at a problem
through a Different Lens
 A global pharmaceutical leader in medical devices, sustainable plastics and protective materials needed controls expertise to complete an Interlocks and Alarms IOQ on a distillation reactor system that creates a liquid used in contact lens manufacturing. Learn how Avanceon provided assistance with not only PLC and HMI programming, but also our electrical, mechanical, project management, and validation expertise to drive the project to a full completion.
A global pharmaceutical leader in medical devices, sustainable plastics and protective materials needed controls expertise to complete an Interlocks and Alarms IOQ on a distillation reactor system that creates a liquid used in contact lens manufacturing. Learn how Avanceon provided assistance with not only PLC and HMI programming, but also our electrical, mechanical, project management, and validation expertise to drive the project to a full completion.Read More
A Shot in the Arm for the System
 When you’re producing vaccines, you don’t want anything to get in the way. See how one major vaccine manufacturer turned to Avanceon to keep production healthy.
When you’re producing vaccines, you don’t want anything to get in the way. See how one major vaccine manufacturer turned to Avanceon to keep production healthy.Read More
Emergency Support Scenarios and Examples: Getting by with a little help from your friends
 Whether through our Automation as a Service offering or our 24 X 7 Emergency Support Services, we have the resources to keep your facility up and running. Here are some examples where we were able to help our customers.
Whether through our Automation as a Service offering or our 24 X 7 Emergency Support Services, we have the resources to keep your facility up and running. Here are some examples where we were able to help our customers.Read More Watch Video
Brownfield SCADA
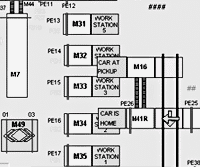 Implementing a SCADA solution to transform your plant into a more modern and efficient production facility.
Implementing a SCADA solution to transform your plant into a more modern and efficient production facility.
When one of North America’s largest beverage manufacturers sought to develop and implement a standardized control system across multiple plants, they turned to Avanceon for support. Learn how the Avanceon team was able to develop and implement a solution; completing the project ahead of deadline in a zero downtime production environment.
Read More
Reclaimed Baking Heat
 Reducing waste, improving quality, saving money and the environment in one fell swoop.
Reducing waste, improving quality, saving money and the environment in one fell swoop.Often times there is more than one solution to a problem. Struggling with inefficiencies and waste during the manufacturing process, one of Avanceon’s long-term partners reached out to us for support with implementing a solution. Bringing a fresh set of eyes to the problem, learn how the Avanceon team was able to identify an alternative solution that was simpler, more efficient and generated significant savings for the facility.
Read More
Software-Based Serialization and Tracking for Pharmaceutical Packaging
 Now that the FDA is officially enforcing the Drug Supply Chain Security Act (DSCSA), many manufacturers are scrambling to implement serialization and track and trace solutions that meet the new requirements. Learn how, after a failed solution from another company, Avanceon was called in to design and implement a successful serialization solution that utilized existing hardware.
Now that the FDA is officially enforcing the Drug Supply Chain Security Act (DSCSA), many manufacturers are scrambling to implement serialization and track and trace solutions that meet the new requirements. Learn how, after a failed solution from another company, Avanceon was called in to design and implement a successful serialization solution that utilized existing hardware.Read More
Effective Validation
 What do you do, when you realize the equipment and processes that make up your new production line weren’t designed to meet FDA requirements? Learn how Avanceon was able to coordinate and document the rigorous validation process for one of our customers, all while meeting a demanding project deadline.
What do you do, when you realize the equipment and processes that make up your new production line weren’t designed to meet FDA requirements? Learn how Avanceon was able to coordinate and document the rigorous validation process for one of our customers, all while meeting a demanding project deadline.Read More
Recipe and Data Management
 What can you do, when you realize your recipe and data collection system have grown so complicated, that they’re no longer practical? Working with a specialized battery manufacturer, Avanceon was able to design and deliver a streamlined solution that standardized the process, increased efficiency and eased troubleshooting.
What can you do, when you realize your recipe and data collection system have grown so complicated, that they’re no longer practical? Working with a specialized battery manufacturer, Avanceon was able to design and deliver a streamlined solution that standardized the process, increased efficiency and eased troubleshooting.Read More
Modifying Existing CIP Systems
 Recent innovations in CIP technology allow manufacturers to save money and reduce waste while still conforming to food safety and product quality standards. Learn how Avanceon was able to develop a custom solution to modify an existing CIP system and how our customer has benefitted from greater flexibility and cost savings.
Recent innovations in CIP technology allow manufacturers to save money and reduce waste while still conforming to food safety and product quality standards. Learn how Avanceon was able to develop a custom solution to modify an existing CIP system and how our customer has benefitted from greater flexibility and cost savings.Read More Watch Video
